Does Stainless Steel Rust?
In general, stainless steel does not rust, but if it is used or maintained improperly, or in a harsh environment, it will rust or tarnish, for example, the photo above shows a stainless steel shackle on a cruise ship corrodes due to long-term exposure to the marine environment, the surface appears brown rust and spots.
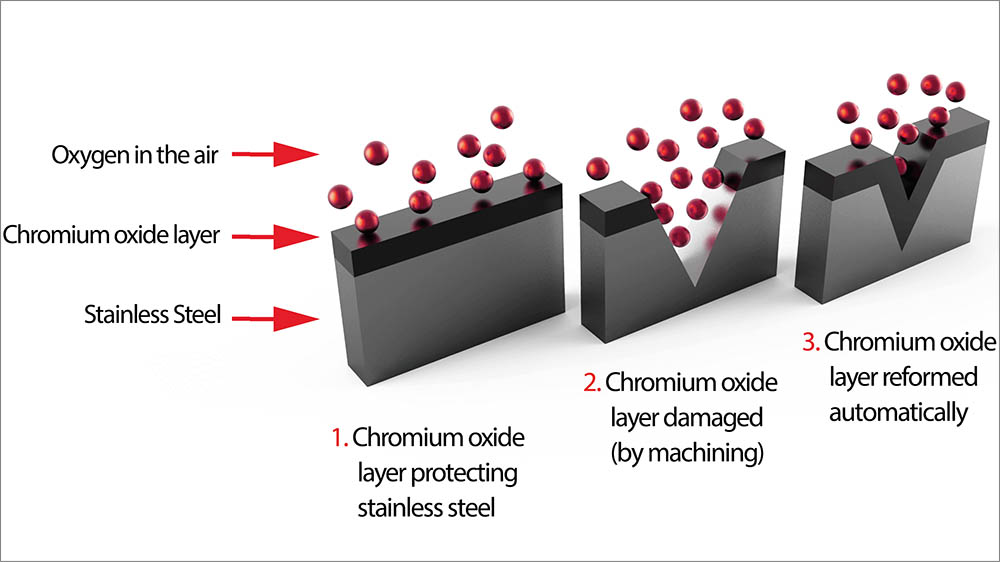
The reason why stainless steel does not rust is because it contains chromium (Cr) and nickel (Ni). These two metal elements will form a dense oxide film with oxygen, this layer of oxide is also called a passivation film, which enhances the corrosion resistance. When the surface of stainless steel is scratched, it will instantly form a new oxide film, thus preventing it from further rusting and corrosion.
Why Does Stainless Steel Rust or Tarnish?
Stainless steel rust because the oxide film on its surface is continuously destroyed, therefore loses its rust proof effect, resulting in the continuous oxidation of iron.
What Causes Stainless Steel to Rust?
Halides are one of the most serious factors that cause stainless steel to rust, they can destroy the passivation film. Halides include chloride, fluoride, bromide, iodide and some inter-halogen compounds. Among them, chloride is the most common in nature and daily life. For example, chlorine ions (Cl-) are widely present in salt, sauce, physiological saline, bleach, disinfectant, salt water lake, and sea water, etc. If stainless steel is polluted by these factors for a long time and is not cleaned in time, it will rust or tarnish.
Other factors that can cause stainless steel to rust include: long-term exposure to inorganic acids such as nitric acid, sulfuric acid, hydrochloric acid, hydrofluoric acid, etc. Organic acids such as acetic acid, formic acid, etc. Alkaline such as sodium hydroxide (NaOH), etc.
Different Grades of Stainless Steel Have Different Corrosion Resistance
Different grades of stainless steel have different rust resistance due to different Cr-Ni content. Other chemical elements that affect corrosion resistance are: contents of carbon (C), phosphorus (P), sulfur (S), molybdenum (Mo), titanium (Ti), copper (Cu), niobium (Nb), etc.
According to the organization methods, they can be divided into 4 classifications:
- Austenite: 200 (essentially Cr-Mn alloys) and 300 series (essentially Cr-Ni alloys), for example, 201, 302, 304 (18/8 stainless steel), 310, 316, 317, 321, etc.
- Ferrite: essentially Fe-Cr alloys, 400 series, such as 430
- Martensite: essentially Fe-Cr-C alloys, 400 series, such as 410, 420, 440, etc.
- Duplex stainless steel: including 2204, 2205, 2507, etc.
In terms of chloride corrosion resistance, the rust resistance ranking of common stainless steel grades is as follows:
904L > 2205 > 316L > 316 > 304L > 304 > 302 > 201 > 430 > 440.